About Us
Regenerative medicine business of Teijin Group
Teijin Group's purpose is "Pioneering solutions together for a healthy planet", and we will achieve our Long-Term Vision: "To be a company that supports the society of the future" by being a company that prioritizes the health of the planet, protects the environment, and supports a circular society, as well as resolve issues for patients, families, and communities in need of greater support.
In accordance with the commitment, Teijin group offers Contract Development and Manufacturing Organization (CDMO) services through the group companies “TEIJIN REGENET Co., Ltd.” and “Japan Tissue Engineering. Co., Ltd. (J-TEC)”. TEIJIN REGENET and J-TEC specialize in Cell & Gene therapy (CGT) including tissue-engineered products, with the aim of supporting the process development and manufacturing of CGT products in Japan - taking innovative science from concept to patient treatment.



- TEIJIN REGENET Co., Ltd.
-
Teijin Regenet was established on August 1, 2023, as a wholly owned subsidiary of Teijin through a company split, succeeding the rights and obligations related to Teijin’s regenerative medicine CDMO business. This corporate reorganization enables Teijin Regenet to execute its regenerative medicine CDMO strategy with greater agility and flexibility. Furthermore, under a licensing agreement between Teijin and J-TEC executed in April 2023, Teijin Regenet delivers well-established CDMO services by leveraging J-TEC’s expertise and know-how.


- Japan Tissue Engineering Co., Ltd.
-
Japan Tissue Engineering (J-TEC) is a regenerative medicine manufacturer with the vision of "Creating a Future for Regenerative Medicine" and has been a member of the Teijin Group since March 2021. As a pioneer of regenerative medicine in Japan, J-TEC has obtained marketing authorization of the following regenerative medical products.
- October, 2007 : Autologous cultured epidermis "JACE ®" (Japan's first regenerative medicine product)
- July, 2012 : Autologous cultured cartilage "JACC ®"
- March, 2020 : Autologous cultured corneal epithelium "NEPIC ®"
- June, 2021 : Autologous cultured oral mucosal epithelium "OCURAL ®"
- March, 2023 : Autologous cultured epidermis containing melanocyte "JACEMIN ®"
Of the 21 regenerative medicine products currently approved in Japan, five are J-TEC products (as of November 2025).
TEIJIN CDMO's strength
Global Infrastructure and Strategic Partnerships Supporting Cell & Gene Therapy / Regenerative Medicine *As of November 2025
- Through strategic partnerships with TFBS Bioscience, Inc. (Taiwan) and Mediridge Co., Ltd. (Japan), we have established a robust viral vector supply framework and a comprehensive range of related testing services, supporting accelerated development of ex vivo gene therapies.
- Through strategic collaborations with leading regenerative medicine CDMOs—including National RESILIENCE (USA), Hilleman Laboratories (Singapore), and Cell Therapies (Australia)—we have built a global manufacturing platform compliant with international regulatory and quality standards. This enables the commercialization and supply of regenerative medicine products not only in Japan, but also with a view toward global market expansion.

"Kashiwa-no-ha Facility", TEIJIN REGENET
- "Kashiwa-no-ha Facility" serves as a core manufacturing function in Kashiwa-no-ha regenerative medicine platform in collaboration with National Cancer Center, Mitsui-Fudosan, and J-TEC. This platform provides one-stop service from early research and development to manufacturing of clinical supply and business strategy with seed holders of Cell & Gene Therapy / Regenerative Medicine, so that we can contribute to producing innovative medical solutions for the diseases with unmet medical needs.
- The site is adjacent to the National Cancer Center East Hospital and it enables to provide geographical and functional benefits for the R&D consortium.
"Iwakuni Factory", TEIJIN REGENET
- "Iwakuni Factrory" provide the integrated manufacturing services through the seamless linkage with Kashiwa-no-ha Facility, from scale-up/scale-out of clinical supply manufacturing to commercial supply manufacturing. We can support various needs of manufacturing such as gene transduction, mass-scale cell culture, and tissue culture.
- The Teijin Group has experience in developing unique pharmaceuticals and medical devices in-house, establishing high-quality production technologies, and commercial supply chain in Japan and overseas. We provide commercial manufacturing services by fully utilizing well established know-hows.
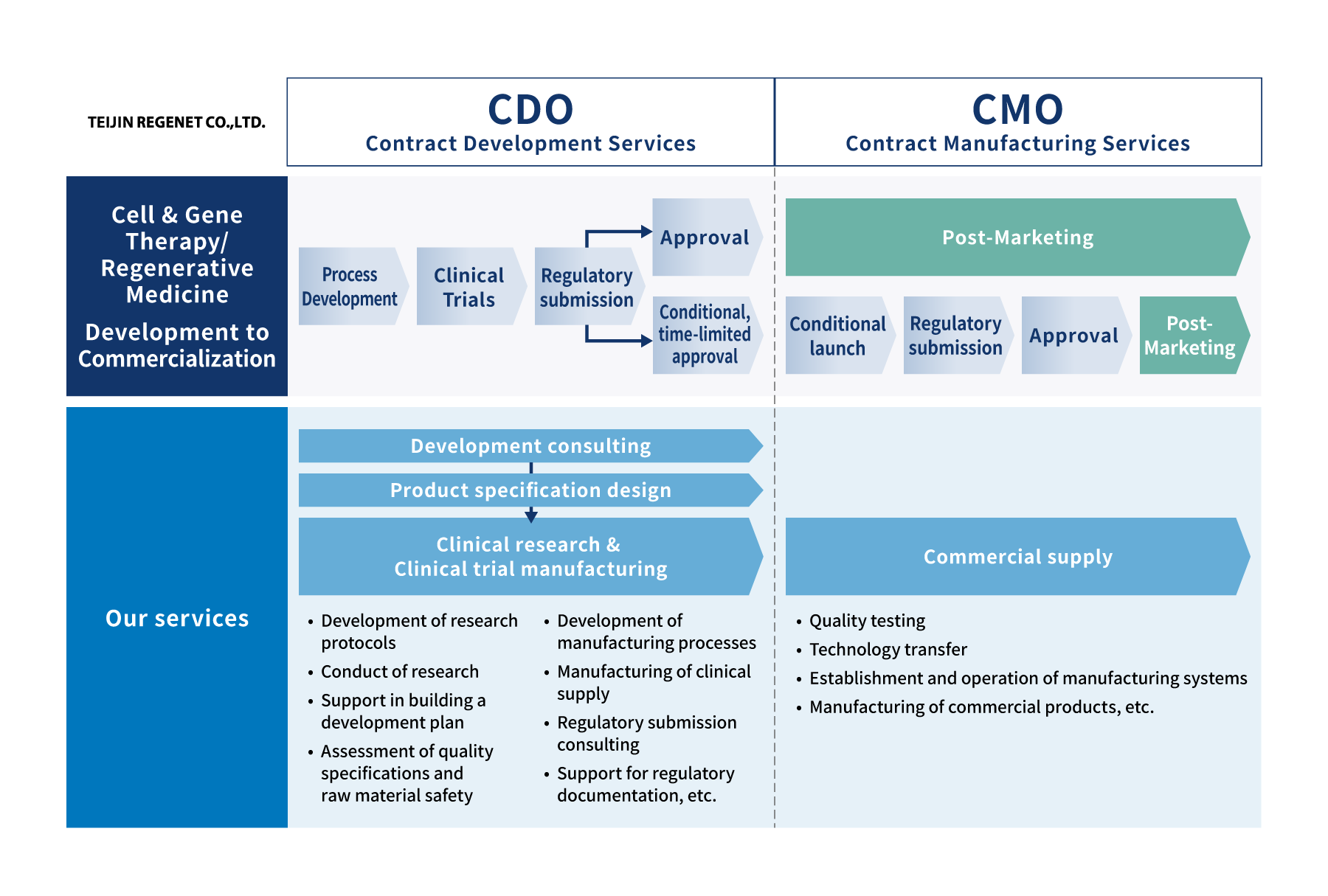
One-stop solution for Cell & Gene Therapy / Regenerative Medicine from development to commercialization
A One-Stop Ecosystem for Regenerative Medicine Development and Commercialization, Powered by Strategic Partnerships
- In July 2025, Teijin Regenet, in collaboration with Toho Holdings and ITOCHU Corporation, established a Regenerative Medicine Ecosystem that provides end-to-end, one-stop support—from R&D through commercialization—for regenerative medicine products.
Through this ecosystem, biotech ventures can efficiently access high-quality, integrated services, enabling accelerated development timelines and earlier market entry.

Japan Tissue Engineering Co., Ltd. (Located Gamagori)
Regulatory approval of the five regenerative medicine products, the largest number in Japan *As of November 2025
- As a pioneer in regenerative medicine, we have cultivated our capability of development, production, and sales, and established the mutual trust relationships of with the regulatory authorities in Japan through the approvals of the multiple products.
- Of the 21 regenerative medicine products in Japan, J-TEC has been making 5 regulatory approvals, including the first and second regenerative medical products in Japan. This is the highest number of achievements of regulatory approvals in Japan.
A track record of more than 260 deals *As of the end of March 2025
We have experience 264 deals (number of the contracts), mainly in academia and bio-ventures in Japan.

Our Manufacturing Expertise in various types of cells and vectors
(Below are representative examples)
Immune cells
- T-cells (CAR-T cells, etc.)
- Dendritic cells
- Lymphocytes
- Monocytes
Stem cells
- Mesenchymal Stromal (derived from bone marrow, adipose tissue, and synovium)
- iPS cells
(iPS cell-derived retinal pigment epithelial cells, iPS cell-derived intestinal epithelial cells, etc.)
Somatic cells
- Epithelial cells
- Pigment cells
- Chondrocytes
Vectors
- Lentiviruses
- Retroviruses
- Plasmids

